Results for ""
Breast cancer is among the leading cancers in India, and among the top 5 causes of cancer-induced mortality among women world over. NIRAMAI's pain-free, touch-free breast cancer detection, driven and augmented with AI and ML, is a revolutionary technology for early screening.
Deeptech healthcare company NIRAMAI's Thermalytix™ Solution has received the prestigious CE Mark Approval, ISO 13485 and MDSAP (Medical Device Single Audit Program) International Certifications. The CE mark Approval indicates that the product may be sold freely in any part of the European Economic Area. The ISO 13485 and MDSAP certifications endorse medical device manufacturers for their compliance with the international medical device quality standards and regulatory requirements.
The Thermalytix™ Solution and quality management processes were audited by an international notified body consisting of expert auditors from India, Germany and Australia. This CE mark approval shows that the product meets EU requirements for medical devices under MDD93/42/EEC for safety, and performance. With these quality certifications, NIRAMAI‘s Thermalytix™ solution can now be applied to all countries of Europe, as well as applicable to many Asian, African and Middle East countries, said the company in a statement.
Dr. Geetha Manjunath, Founder and CEO of NIRAMAI comments, “We are delighted to receive the CE marking, ISO and MDSAP certifications for our technology, further strengthening our resolve to detect breast cancer in early stages and save lives. These certifications enable us to expand our operations internationally and meet the strong global demand we are seeing for our novel solution."
In the future, NIRAMAI plans to collaborate with renowned radiologists of UK's National Health Service (NHS) and other key opinion leaders for an initial market testing in Europe, Africa and Asia. In addition, they are in the process of partnering with more Indian Healthcare providers for pan-India adoption.
Manish Singhal, Founding Partner of pi Ventures and lead investor of NIRAMAI said, "NIRAMAI's unique approach of detection of breast cancer using AI and thermal is a game-changer on how lives can be saved from breast cancer. With the ISO and CE certification, now Thermalytix can be made available to women beyond India especially in entire Europe, Middle East and several countries in Southeast Asia leading to global impact. We are super excited to take this technology to the world."
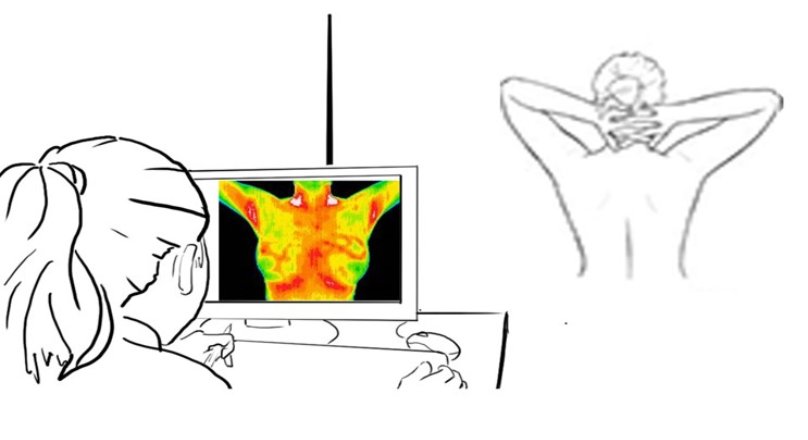
NIRAMAI Thermalytix™ test is revolutionizing breast health screening by enabling privacy-aware, safe and accurate breast health checks in hospitals, in screening camps and also at homes. NIRAMAI Test is already being used in renowned hospitals and clinics across various Indian cities, including HCG Hospital, Apollo Clinics, HealthSpring Diagnostics, among others.
NIRAMAI Thermalytix™ solution uses a high-resolution thermal sensing device to capture thermal images and a cloud-hosted analytics solution for analysing these thermal images using proprietary machine learning-based software for reliable, early and accurate breast cancer screening. The test has been clinically validated to show better sensitivity than mammography.
WATCH: Conversations with INDIAai ft. Geetha Manjunath, CEO & Cofounder of NIRAMAI